Medicinal Chemistry of Herbal Flower Nelumbo Nucifera Gaertn
Sai Vuyyuru

The herbal flowers of Nelembu Nucifera Geratn were used in Ayurvedic, Unani & Sidha ancient cultures for various medical conditions such as dysentery, diarrhea, piles, dyspepsia and ringworm without full understandings of the medicinal chemistry of this herbal plant.
Nelumbo Nucifera Gaertn consists of dried flowers of Nelumbo nucifera Gaertn. Syn. Nelumbium speciosum Willd. Fam. Nymphaeaceae; a large, aquatic herb with submerged rhizome, cultivated throughout warmer parts of India upto an altitude of 1000 m.
OTHER NAMES
San. - Abja; Aravinda; Pankaja; Utpala
Assam. - Podum
Bengal - Padma phool; Salaphool
Eng. - Lotus
Guj. - Kamal
Hindi - Kamal; Kanwal
Kan. - Kamal; Tavare; Naidile; Tavaregedd
Mal. - Komala; Thamara; Santhamara, Centamarai
Ori. - Padma
Punj. - Kanwal; Pamposh
Tam. - Tamarai; Centamarai
Tel. - Kaluva; Tamarapuvvu
Urdu - Kamal
DESCRIPTION
Drug occurs as entire shriveled yellowish brown pieces of flowers, 10 to 15 cm in diameter, comprising of calyx, corolla, androecium, gynoecium, thalamus and pedicel; sepals leaf-like, 3 to 5 cm long, 1.3 to 2 cm wide, dark brown, broken; petals numerous, elliptic, obtuse, membranous, finely veined, 2 to 4 cm long, 1.2 to 2 cm wide, yellowish-brown; anther, erect, linear 1.4 to 2 cm long, extended into clavate appendages; gynoecium apocarpous; carpels many, free, embedded in a creamy, topped fleshy thalamus (torus) 3-5 cm long and 2.5 to 3 cm wide; style minute, stigma terminal, ovary single celled, ovule solitary, suspended, anatropus. (Plate-1)
Microscopy
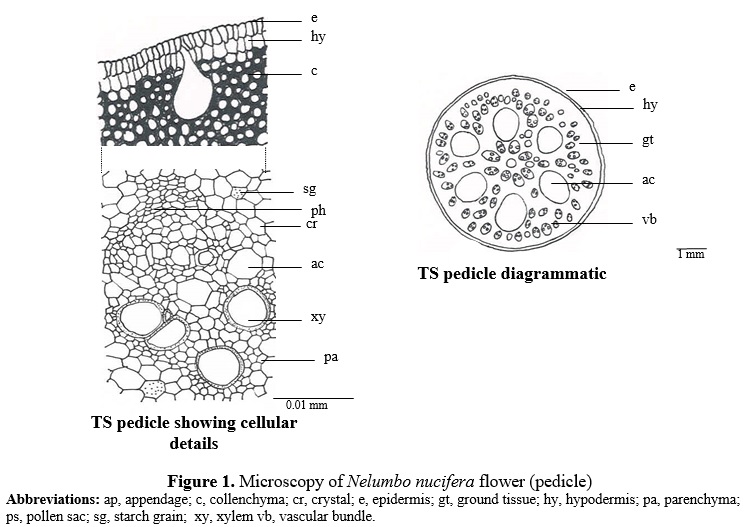
TS of pedicel shows two layered epidermis consisting of radially elongated cells. Underneath this lies 6 to 8 layered collenchymatous hypodermis embedded with bases of pear shaped air cavities its narrow and being connected with cells of epidermis, the remaining ground tissue is parenchymatous, traversed with conjoint, collateral vascular strands and circular to oval huge air cavities. Small air cavities traverse throughout the phloem tissue of the vascular bundles also simple starch grains, prismatic and cluster crystals of calcium oxalate are present throughout the parenchymatous cells of the ground tissue. (Fig. 1)
TS of pedicel shows two layered epidermis consisting of radially elongated cells. Underneath this lies 6 to 8 layered collenchymatous hypodermis embedded with bases of pear shaped air cavities its narrow and being connected with cells of epidermis, the remaining ground tissue is parenchymatous, traversed with conjoint, collateral vascular strands and circular to oval huge air cavities. Small air cavities traverse throughout the phloem tissue of the vascular bundles also simple starch grains, prismatic and cluster crystals of calcium oxalate are present throughout the parenchymatous cells of the ground tissue. (Fig. 1)
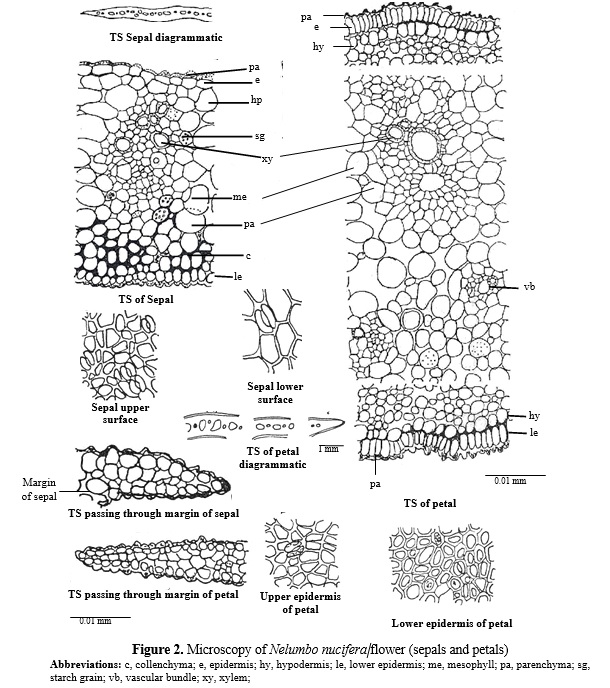
The transverse section of sepal is broad centrally and has thin narrow margins. The cells of lower and upper epidermis are papillate. In surface view these are straight walled and angular, the anomocytic stomata are present on both the surfaces. On upper side twin stomata are also observed. The single layered hypodermis is followed by loosely arranged mesophyll cells. Number of collateral vascular bundles are scattered in mesophyll region. Starch grains and large air cavities are present in mesophyll region and phloem parenchyma, margin of sepals consists of 1-2 layers of loosely arranged parenchymatous cells. (Fig.2).
TS of petals are almost similar to sepals except the cells of upper and lower epidermis are highly papillate and thick walled, stomata are present on both the surfaces and are sunken and anomocytic type,cluster crystal of calcium oxalate are present in epidermal cells. (Fig. 2).
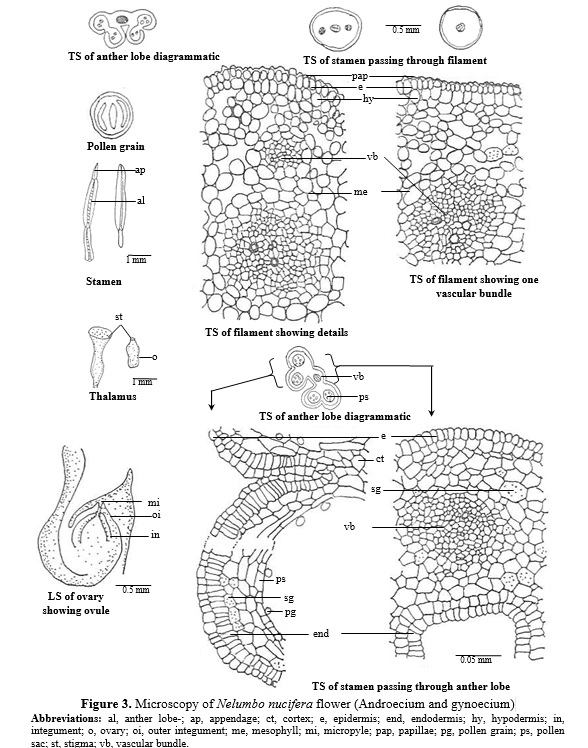
Androecium – A majority of the stamens have clavate shaped distal sterile appendages with obtuse apex. Four slender long, sublateral, microsporangia occur in pairs. A transverse section of the filament and appandage appears almost circular in outline covered by single layered papillate epidermis followed by single layered hypodermis which is filled with reddish brown contents, followed by loosely arranged parenchymatous mesophyll. The vascular supply varying from 1-5 bundles. In the basal region large central bundle flanked by 4 lateral bundles. (Fig. 3).
Androecium – A majority of the stamens have clavate shaped distal sterile appendages with obtuse apex. Four slender long, sublateral, microsporangia occur in pairs. A transverse section of the filament and appandage appears almost circular in outline covered by single layered papillate epidermis followed by single layered hypodermis which is filled with reddish brown contents, followed by loosely arranged parenchymatous mesophyll. The vascular supply varying from 1-5 bundles. In the basal region large central bundle flanked by 4 lateral bundles. (Fig. 3).
TS passing through microsporangia shows a single layered epidermis followed by a radially elongated endothecial layer followed by 4-5 celled parenchymatous cells. The vascular supply is reduced to single bundle. (Fig. 3)
Pollen grains – Pollen grains are 2-4 zonocolporate, size 72 x 66 mm (55.40 – 72 X 44.32 – 62 mm) in diameter, exine thicker than nexine, tectate – punctate. (Fig. 3)
Powder
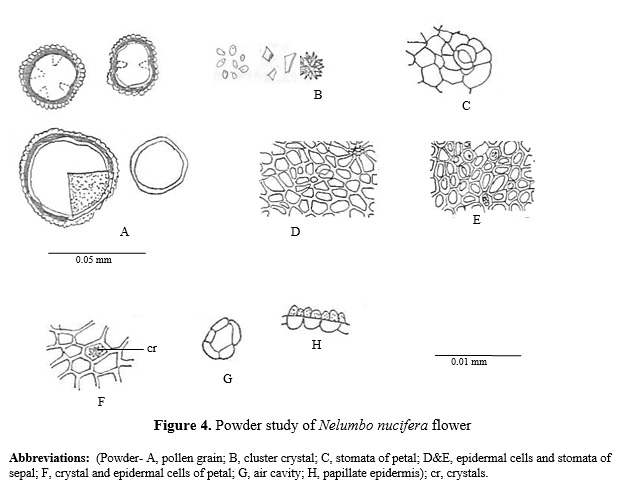
Powder light brown in colour, sweet in taste with no characteristic odour, on microscopical examination it shows rosette and prismatic crystals of calcium oxalate, pieces of papillate epidermis, aircavities, pollengrain, vessels, epidermal cells with stomata of petals and sepals. (Fig.4).
CHEMICAL CONSTITUENTS
Major
Gallic acid (No supporting literature available till date)
Major
Gallic acid (No supporting literature available till date)
Others
Linalool2, palmitic acid2, 1,4-dimethoxybenzene2. myristic acid2, ß-pinene2, limonene2, trans-ocimenene2, -terpinene2, ß-caryophyllene2, -humulene2, naphthalene2, methyl naphthalene2, dimethyl naphthalene2, n-tetradecane2, n-pentadecane2, n-hexadecane2, n-heptadecane2, n-octadecane2, n-nonadecane2, n-eicosane2, n-heneicosane2, n-docosane2, n-tricosane2, n-tetracosane2, n-pentacosane2, n-hexacosane2, n-heptacosane2, n-octacosane2, n-nonacosane2, n-triacontane2, n-hentriacontane2, n-dotriacontane2, n-tritriacontane2, n-hexadecane2, n-nonadecane2, n-eicosane2, n-tricosane2, 1,8-cineole2, caryophyllene oxide2, -cadinol2, terpinen-4-ol2, -terpineol2, geranyl linalool2, phytol2, methyl propyl phenol2, cis-jasmone2, methyl palmitate2, methyl stearate2, methyl eicosanoate2, methyl tricosanoate2, ethyl hexanoate2, ethyl stearate2, linalyl hexanoate2.
Linalool2, palmitic acid2, 1,4-dimethoxybenzene2. myristic acid2, ß-pinene2, limonene2, trans-ocimenene2, -terpinene2, ß-caryophyllene2, -humulene2, naphthalene2, methyl naphthalene2, dimethyl naphthalene2, n-tetradecane2, n-pentadecane2, n-hexadecane2, n-heptadecane2, n-octadecane2, n-nonadecane2, n-eicosane2, n-heneicosane2, n-docosane2, n-tricosane2, n-tetracosane2, n-pentacosane2, n-hexacosane2, n-heptacosane2, n-octacosane2, n-nonacosane2, n-triacontane2, n-hentriacontane2, n-dotriacontane2, n-tritriacontane2, n-hexadecane2, n-nonadecane2, n-eicosane2, n-tricosane2, 1,8-cineole2, caryophyllene oxide2, -cadinol2, terpinen-4-ol2, -terpineol2, geranyl linalool2, phytol2, methyl propyl phenol2, cis-jasmone2, methyl palmitate2, methyl stearate2, methyl eicosanoate2, methyl tricosanoate2, ethyl hexanoate2, ethyl stearate2, linalyl hexanoate2.
TLC IDENTITY TEST
Test solution:
Reflux 5 g of the powdered drug with methanol on a water bath for 30 min. Filter and remove the solvent under reduced pressure consecutively three times, dissolve 25 mg of the residue in 10 ml of methanol. Use this solution for TLC profile.
Standard solution:
Dissolve 10 mg of gallic acid in 10 ml of methanol.
Solvent system:
Toluene: Ethyl acetate: Formic acid (5:5:1)
Procedure:
Apply 10 l each of the test and standard solutions on a precoated silica gel 60 F254 TLC plate (E. Merck) of uniform thickness 0.2 mm. Develop the plate in the solvent system in twin trough chamber to a distance of 8 cm.
Apply 10 l each of the test and standard solutions on a precoated silica gel 60 F254 TLC plate (E. Merck) of uniform thickness 0.2 mm. Develop the plate in the solvent system in twin trough chamber to a distance of 8 cm.
Visualization:
Observe the developed plate under UV light at 254 (Plate 2A) and 366nm (Plate 2B).and under visible light after derivatization with anisaldehyde-sulfuric acid reagent and heat the plate at 110C for 10 min. (Plate 2C). Record the Rf values and colour of the resolved bands (Table 1).
Observe the developed plate under UV light at 254 (Plate 2A) and 366nm (Plate 2B).and under visible light after derivatization with anisaldehyde-sulfuric acid reagent and heat the plate at 110C for 10 min. (Plate 2C). Record the Rf values and colour of the resolved bands (Table 1).
Evaluation:
A band (Rf= 0.55) corresponding to gallic acid is visible in both reference solution and test solution tracks.
A band (Rf= 0.55) corresponding to gallic acid is visible in both reference solution and test solution tracks.

ASSAY / ANALYTICAL METHOD
TLC densitometric estimation of gallic acid.
TLC plates
Precoated silica gel 60 F254 (E. Merck) of 0.2 mm thickness.
Precoated silica gel 60 F254 (E. Merck) of 0.2 mm thickness.
Solvent system
Toluene: Ethyl acetate: Formic acid (5: 5: 1)
Scanning
Under UV light at 278 nm.
Under UV light at 278 nm.
Test solutions 1 ,2 and 3 are from samples collected from Bangalore, Cochin and Imphal respectively.
Test solution:
Reflux 5 g of the finely powdered drug with 25ml of methanol on a water bath for 25 min consecutively three times, filter and remove the solvent under reduced pressure. Dissolve 25 mg of the extractive in 20 ml of methanol. Use this portion for estimation of the gallic acid in the drug.
Test solution:
Reflux 5 g of the finely powdered drug with 25ml of methanol on a water bath for 25 min consecutively three times, filter and remove the solvent under reduced pressure. Dissolve 25 mg of the extractive in 20 ml of methanol. Use this portion for estimation of the gallic acid in the drug.
Standard solution:
Dissolve 10 mg of gallic acid in 10 ml of methanol from this stock solution prepare standard solutions of 20-100 mg/ml by transferring aliquots (0.2 to 1 ml) of stock solution to 10 ml volumetric flask and adjusting the volume to 10 ml with methanol.
Dissolve 10 mg of gallic acid in 10 ml of methanol from this stock solution prepare standard solutions of 20-100 mg/ml by transferring aliquots (0.2 to 1 ml) of stock solution to 10 ml volumetric flask and adjusting the volume to 10 ml with methanol.
Calibration curve:
Apply 10 ml of each of the standard solutions (200-1000 ng per spot) on a precoated silica gel 60 F254 TLC plate. Develop the plate in the solvent system in twin trough chamber to a distance of 8 cm, dry it and scan densitometrically at 278 nm, record the peak area and prepare the calibration curve by plotting peak area against concentration of the gallic acid applied.
Apply 10 ml of each of the standard solutions (200-1000 ng per spot) on a precoated silica gel 60 F254 TLC plate. Develop the plate in the solvent system in twin trough chamber to a distance of 8 cm, dry it and scan densitometrically at 278 nm, record the peak area and prepare the calibration curve by plotting peak area against concentration of the gallic acid applied.
Estimation of gallic acid in the drug:
Apply 10 ml of the test and standard solutions in triplicate on a precoated TLC plate (E. Merck) of uniform thickness of 0.2 mm. Develop the plate in the solvent system and record the peak area of gallic acid as described above for the calibration curve. Calculate the amount of gallic acid present in the sample from calibration curve of gallic acid. (Fig.2).
Apply 10 ml of the test and standard solutions in triplicate on a precoated TLC plate (E. Merck) of uniform thickness of 0.2 mm. Develop the plate in the solvent system and record the peak area of gallic acid as described above for the calibration curve. Calculate the amount of gallic acid present in the sample from calibration curve of gallic acid. (Fig.2).
The percentage of gallic acid ranges from 0.85 to 0.95 (w/w) in the samples analyzed.
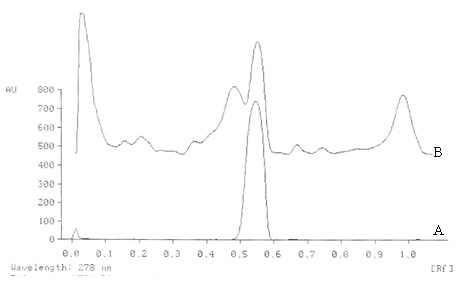
Figure 2. TLC densitometric scan at 278 nm of test solution of methanolic extract of Nelumbo nucifera flowers. A. Gallic acid standard B. Test solution
QUANTITATIVE STANDARDS
Foreign matter: Not more than 2 per cent. Moisture content: Not more than 5 per cent. Total ash: Not more than 6 per cent. Acid-insoluble ash: Not more than 1 per cent. Ethanol-soluble extractive: Not less than 16 per cent. Water-soluble extractive: Not less than 30 per cent.
ADULTERANTS/ SUBSTITUTES
The petals and whole flowers of Nymphaea species can be adulterated or substituted, the petals and sepals of Nelumbo nucifera can be differentiated by the absence of sclerieds.
PHARMACOLOGY
The aqueous and alcoholic extracts of the flowers showed anti-hypoglycemic activity in rabbits3 . On chronic treatment there was no sustained suppression of fasting blood sugar level4. The ethanolic extract of the seeds reported to have significant antioxidant and hepatoprtective activities5.
The aqueous and alcoholic extracts of the flowers showed anti-hypoglycemic activity in rabbits3 . On chronic treatment there was no sustained suppression of fasting blood sugar level4. The ethanolic extract of the seeds reported to have significant antioxidant and hepatoprtective activities5.
THERAPEUTIC CATEGORY
Haemorrhagic disorders6.
Haemorrhagic disorders6.
SAFETY ASPECTS
Drug is traditionally considered safe with in prescribed dose6.
Drug is traditionally considered safe with in prescribed dose6.
DOSAGE
12-24 g of the drug for decoction6.
12-24 g of the drug for decoction6.
REFERENCES
- Mehrotra S, Shome U, Sharma, HP. Pharmacognostic studies on the flower of Nelumbo nucifera Gaertn. New Botanist 1987; 14: 126-146.
- Omata A, Yomogida K, Nakamura S,Ohta T, Izawa Y, Watanabe S. The scent of lotus flowers. J Ess Oil Res 1991; 3:221-227.
- Huralikuppi JC, Christopher AB, Stephen PM. Anti-diabetic effect of Nelumbo nucifera (Gaertn): Part I preliminary studies in rabbits. Phytothera Res 1991; 5: 54-58.
- Huralikuppi JC, Cristopher AP, Stephen PM. Antidiabetic effect of Nilumbo nucifera Gaertn part II. Phytother Res 1991; 5: 217-123.
- Sohn DH, Kim YC, Oh SH, Park EJ, Li X, Lee BH. Hepatoprotective and free radical scavenging effects of Nelumbo nucifera. Phytomed 2003; 10: 165-169.
- The Ayurvedic Pharmacopoeia of India. Part-I Vol II, 1st Edition, Govt. of India. Ministry of Health and Family Welfare, Department of ISM&H, New Delhi 1999. p. 69-70.
The article is prepared by Sai Vuyyura, an employee of Cure Discount Pharmacy, the contents of which cannot be reproduced, without the written permission of Cure Pharmacy Inc., which has the sole proprietary interest in this publication.

